ISSUE #14 ABDR NEWSLETTER OUT NOW
December 21, 2020
Welcome to the latest issue of the Australian Breast Device Registry (ABDR) update.
National recruitment
Despite #COVID-19, the ABDR continued to grow in 2020. Our sincere thanks go to Australians undergoing breast device surgery, their surgeons, and health care facilities for supporting this important work. The 290+ participating sites are listed here.
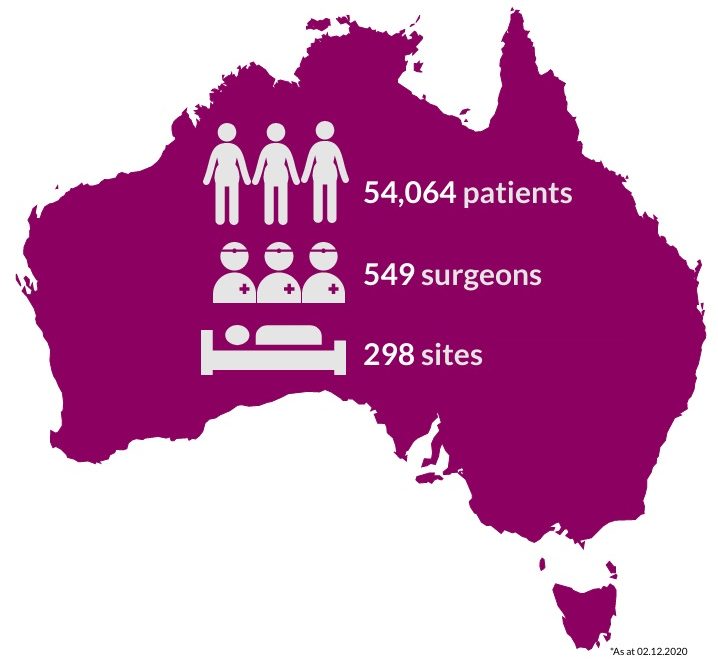
ABDR 2019 Annual Report
- We are pleased to release the 2019 ABDR annual report involving almost 50,000 Australians.
- 11,960 patients with devices were included in the registry during 2019, bringing the cumulative total to 49,563 registered patients.
- Between 2012 – 2019 a total of 10,634 patients had breast reconstructive surgery, comprising 14,912 total procedures, utilising 22,943 breast devices.
- A total of 36,233 patients had aesthetic surgery (cosmetic augmentation), involving 37,852 procedures and 75,155 devices over the same period.
- Requests to reproduce content in this report should be sent to abdr@monash.edu
- View the report here.
ABDR Team news
Farewell
We bid a fond farewell to ABDR Project Lead, Associate Professor Ingrid Hopper in October this year. Ingrid steered the ABDR through a period of enormous growth and expansion between December 2015 – November 2020.
Under her watch, the ABDR published multiple peer reviewed articles, four Annual Reports, implemented the PROMs (Patient Reported Outcome Measures) sub-study and continued to lead the ABDR in the International Collaboration of Breast Registry Activities (ICOBRA).
Ingrid is focussing on clinical work and her interests in heart failure management and research.
The team thank Ingrid for her for leadership and commitment to the registry over the years and wish her all the best for the future.
 Welcome to ABDR’s new lead
Welcome to ABDR’s new lead
We warmly welcome Professor Susannah Ahern to the helm. Susannah is the Head of the Registry Science and Research Program at Monash University, and leads six clinical quality registries.
As a previous medical administrator, she is course Coordinator of the Master of Health Management/Master Health Administration at SPHPM. She is a member of the Australian Commission in Safety and Quality in Health Care’s Advisory Committee for CQRs, the Commonwealth Department of Health’s CQR Strategy Expert Advisory Group and the Victorian DHHS’s CQR Advisory Group. Susannah now represents the ABDR on the TGA Breast Implant Expert Working Group and ICOBRA.
In brief
In October 2020, the TGA announced the cancellation of three breast device products from the Australian market, following suspension of the devices in May.
Most recent publications
- Ng S, Parker E, Pusic A, Farrell G, Moore C, Elder E, Cooter RD, McNeil J, Hopper I. Lessons Learned in Implementing Patient Reported Outcome Measures (PROMs) in the Australian Breast Device Registry (ABDR). Aesthet Surg J. 2020 Dec 17:sjaa376. doi: 10.1093/asj/sjaa376. Epub ahead of print. PMID: 33331907
- Bargon CA, Becherer BE, Young-Afat DA, van Bommel ACM, Hommes J, Hoornweg MJ, Keuter XHA, de Fazio S, Melnikov D, Monton Echeverria J, Perks GAB, Lumenta DB, Couturaud B, von Fritschen U, Stark B, Hölmich LR, Crosbie A, Lispi L, Campanale A, Cooter RD, Pusic AL, Hopper I, Mureau MAM, Rakhorst HA. Moving breast implant registries forward: Are they FAIR and Functional? Journal of Plastic, Reconstructive & Aesthetic Surgery. 2020 Journal pre-proof published online October 17. doi.org/10.1016/j.bjps.
2020.10.001 - Spronk PE, Begum H, Vishwanath S, Crosbie A, Earnest A, Elder E, Lumenta DB, Marinac-Dabic D, Moore CC, Mureau MA, Perks G, Pusic AL, Stark B, von Fritschen U, Klein H, Cooter RD, Rakhorst HA, Hopper I. Toward International Harmonization of Breast Implant Registries: International Collaboration of Breast Registry Activities Global Common Data Set. Plastic and Reconstructive Surgery. 2020 August. Volume 146 – Issue 2 – p 255-267. doi: 10.1097/PRS.0000000000006969
- Several publications are in the pipeline. To view published papers and ABDR annual reports visit dev-abdr-org.pantheonsite.io/publications
Monash closure dates
Monash University and the ABDR will be closed from Wed 23rd December 2020 and will reopen on Thursday 7th January 2021.
We sincerely thank patients, surgeons, health service staff, ABDR clinical leads and the ABDR Steering Committee for your ongoing support for this important registry. Have a safe festive break and we look forward to continuing our work with you in 2021.